Biomateriali
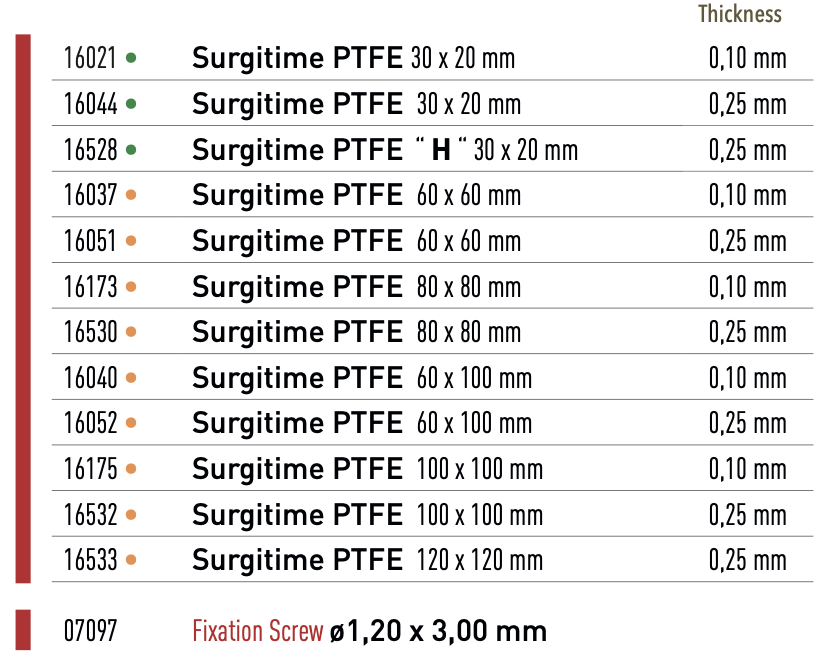
Surgitime PTFE
Nonabsorbable Membrane
 Surgitime PTFE is a nonabsorbable membrane composed of Polytetrafluoroethylene, withthickness of 0.10 or 0.25mm. Surgitime PTFE is 100% biocompatible, syntheticand not of animal origin. It is indicated for in orthopedic, neural, maxillofacial procedures and other medical or dental surgery procedures.
Surgitime PTFE is a nonabsorbable membrane composed of Polytetrafluoroethylene, withthickness of 0.10 or 0.25mm. Surgitime PTFE is 100% biocompatible, syntheticand not of animal origin. It is indicated for in orthopedic, neural, maxillofacial procedures and other medical or dental surgery procedures.
The polytetrafluoroethylene (PTFE) membranes or mechanical barriers for RTG (Guided Tissue Regeneration) has the aim of preventing the migration of epithelial and connective tissue cells, which would cause the inhibition of bone growth, promoting suitable space for the formation of a natural fibrin framework, the bone tissue precursor.
The membrane avoids the tissue competition between the connective tissue and the bone, and has the purpose of isolating the bone grafts promoting tissue regeneration. Surgitime PTFE has selective permeability through its porosity, which enables the nutrition of the fibrin framework and simultaneously preventing the passage of bacteria.
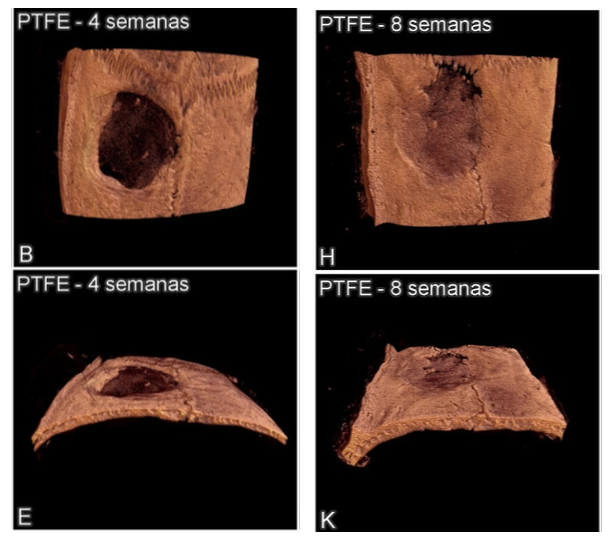
MICRO CT: Morphometry Analysis
Three-dimensional images of the defects created in thecalvaria of mouse: Implanted PTFE membrane.
Scale bar: 2 mm.
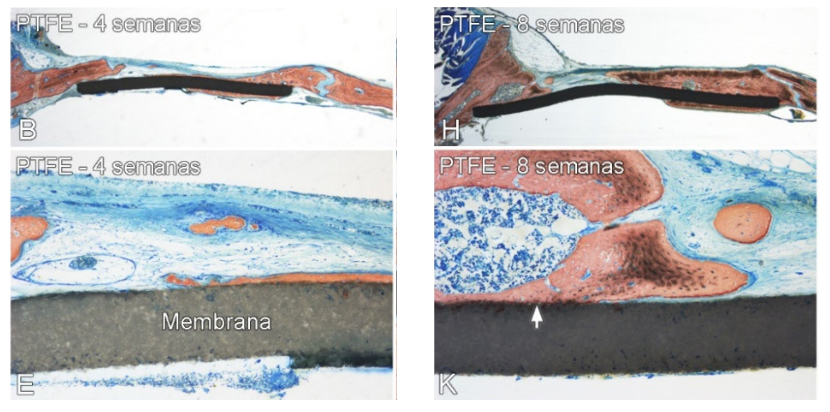
LIGHT MICROSCOPY: Histological Analysis
The newly formed mineralized bone tissue was observed in both sides of the membranes B (4 weeks) and H (8 weeks). The arrows indicate the close contact between bone tissue and the membrane of PTFE (K)
Colour: Stevenel’s blue and Alizarin red
Scale bar: B and H = 2 mm; E and K = 200 micrometers
Features
- High resistance (weight/resistance ratio);
- Chemically inert;
- High chemical resistance in aggressive environments;
- Low inflammability;
- Low coefficient of friction;
- Low dielectric constant;
- Good weathering properties
